Motor variable: Difference between revisions
en>ChrisGualtieri m TypoScan Project / General Fixes, typos fixed: , → , (3) using AWB |
en>Rgdboer m mv refs inline, lk Wirtinger derivative |
||
| Line 1: | Line 1: | ||
{{Use dmy dates|date=July 2013}} | |||
Please find below supplementary chemical data about [[dichloromethane]]. | |||
==MSDS sheets== | |||
The handling of this chemical may incur notable safety precautions. It is highly recommend that you seek the Material Safety Datasheet ([[Material safety data sheet|MSDS]]) for this chemical from a reliable source and follow its directions. | |||
* [http://www2.siri.org/msds/index.php SIRI]{{dead link|date=January 2013}} | |||
* [http://www.sciencestuff.com/msds/C1677.html Science Stuff] | |||
* [http://hazard.com/msds/mf/baker/baker/files/m4420.htm Baker] | |||
* [http://physchem.ox.ac.uk/MSDS/DI/dichloromethane.html Oxford University Chemical Safety Archive]{{dead link|date=January 2013}} | |||
== Structure and properties == <!-- KEEP this header, it is linked to from the infobox on the main article page --> | |||
{| border="1" cellspacing="0" cellpadding="3" style="margin: 0 0 0 0.5em; background: #FFFFFF; border-collapse: collapse; border-color: #C0C090;" | |||
! {{chembox header}} | Structure and properties | |||
|- | |||
| [[Index of refraction]],<ref>{{Cite web|url=http://www.chemicalland21.com/industrialchem/solalc/METHYLENE%20CHLORIDE.htm|title=Methylene Chloride|publisher=chemicalland21.com|accessdate=29 May 2007}}</ref> ''n''<sub>D</sub> | |||
| 1.4242 <!-- Please omit if not applicable --> | |||
|- | |||
| [[Abbe number]] | |||
|? <!-- Please omit if not applicable --> | |||
|- | |||
| [[Dielectric constant]],<ref>''CRC Handbook of Chemistry and Physics'', 47th ed. p E-54</ref> ε<sub>r</sub> | |||
| 9.08 ε<sub>0</sub> at 20°C <!-- Please omit if not applicable --> | |||
|- | |||
| [[molecular dipole moment|Dipole moment]],<ref>http://macro.lsu.edu/howto/solvents/Dipole%20Moment.htm</ref> | |||
| 1.14 D <!-- Please omit if not applicable --> | |||
|- | |||
| [[Bond strength]] | |||
| ? <!-- Specify which bond. Please omit if not applicable --> | |||
|- | |||
| [[Bond length]] | |||
| ? <!-- Specify which bond. Please omit if not applicable --> | |||
|- | |||
| [[Bond angle]] | |||
| ? <!-- Specify which angle, e.g. Cl-P-O. Please omit if not applicable --> | |||
|- | |||
| [[Magnetic susceptibility]] | |||
| ? <!-- Please omit if not applicable --> | |||
|- | |||
| [[Surface tension]]<ref>''CRC Handbook of Chemistry and Physics'', 47th ed. pp F-28 - F-30</ref> | |||
| 26.52 dyn/cm at 20°C | |||
|- | |||
| [[Viscosity]]<ref>''CRC Handbook of Chemistry and Physics'', 47th ed. pp F-33 - F-38</ref> | |||
| 0.449 mPa·s at 15°C<br>0.393 mPa·s at 30°C | |||
|- | |||
|} | |||
== Thermodynamic properties == <!-- KEEP this header, it is linked to from the infobox on the main article page --> | |||
{| border="1" cellspacing="0" cellpadding="6" style="margin: 0 0 0 0.5em; background: #FFFFFF; border-collapse: collapse; border-color: #C0C090;" | |||
! {{chembox header}} | Phase behavior | |||
|- | |||
| [[Triple point]] | |||
| ? K (? °C), ? Pa | |||
|- | |||
| [[Critical point (thermodynamics)|Critical point]]<ref name="cheric_p">{{Cite web|url=http://www.cheric.org/research/kdb/hcprop/cmpsrch.php|title=Pure Component Properties|format=Queriable database|publisher=Chemical Engineering Research Information Center|accessdate=29 May 2007}}</ref> | |||
| 510 K (237 °C), 6100 kPa | |||
|- | |||
| [[Standard enthalpy change of fusion|Std enthalpy change<br/>of fusion]], Δ<sub>fus</sub>''H''<sup><s>o</s></sup> | |||
| +6.160 kJ/mol | |||
|- | |||
| [[Standard entropy change of fusion|Std entropy change<br/>of fusion]], Δ<sub>fus</sub>''S''<sup><s>o</s></sup> | |||
| ? J/(mol·K) | |||
|- | |||
| [[Standard enthalpy change of vaporization|Std enthalpy change<br/>of vaporization]], Δ<sub>vap</sub>''H''<sup><s>o</s></sup> | |||
| +28.6 kJ/mol | |||
|- | |||
| [[Standard entropy change of vaporization|Std entropy change<br/>of vaporization]], Δ<sub>vap</sub>''S''<sup><s>o</s></sup> | |||
| ? J/(mol·K) | |||
|- | |||
! {{chembox header}} | Solid properties | |||
|- | |||
| [[Standard enthalpy change of formation|Std enthalpy change<br/>of formation]], Δ<sub>f</sub>''H''<sup><s>o</s></sup><sub>solid</sub> | |||
| ? kJ/mol | |||
|- | |||
| [[Standard molar entropy]],<br/>''S''<sup><s>o</s></sup><sub>solid</sub> | |||
| ? J/(mol K) | |||
|- | |||
| [[Heat capacity]], ''c<sub>p</sub>'' | |||
| ? J/(mol K) | |||
|- | |||
! {{chembox header}} | Liquid properties | |||
|- | |||
| [[Standard enthalpy change of formation|Std enthalpy change<br/>of formation]], Δ<sub>f</sub>''H''<sup><s>o</s></sup><sub>liquid</sub> | |||
| −124.3 kJ/mol | |||
|- | |||
| [[Standard molar entropy]],<br/>''S''<sup><s>o</s></sup><sub>liquid</sub> | |||
| 174.5 J/(mol K) | |||
|- | |||
| [[Heat capacity]], ''c<sub>p</sub>'' | |||
| 102.3 J/(mol K) | |||
|- | |||
! {{chembox header}} | Gas properties | |||
|- | |||
| [[Standard enthalpy change of formation|Std enthalpy change<br/>of formation]], Δ<sub>f</sub>''H''<sup><s>o</s></sup><sub>gas</sub> | |||
| −95.52 kJ/mol | |||
|- | |||
| [[Standard molar entropy]],<br/>''S''<sup><s>o</s></sup><sub>gas</sub> | |||
| 270.28 J/(mol K) | |||
|- | |||
| [[Heat capacity]], ''c<sub>p</sub>'' | |||
| ? J/(mol K) | |||
|- | |||
| [[van der Waals equation|van der Waals' constants]]<ref>{{Cite web|url=http://www2.ucdsb.on.ca/tiss/stretton/Database/van_der_waals_constants.html|title=van der Waal's Constants for Real Gases|publisher=Tom Stretton's Chemistry Pages|accessdate=29 May 2007}}</ref> | |||
| a = 1244 L<sup>2</sup> kPa/mol<sup>2</sup><br> b = 0.08689 liter per mole | |||
|- | |||
|} | |||
==Vapor pressure of liquid== | |||
{| border="1" cellspacing="0" cellpadding="6" style="margin: 0 0 0 0.5em; background: white; border-collapse: collapse; border-color: #C0C090;" | |||
|- | |||
| {{chembox header}} | '''P in mm Hg''' || 1 || 10 || 40 || 100 || 400 || 760 | |||
|- | |||
| {{chembox header}} | '''T in °C''' || –70.0 || –43.3 || –22.3 || –6.3 || 24.1 || 40.7 | |||
|} | |||
Table data obtained from ''CRC Handbook of Chemistry and Physics'' 47th ed. | |||
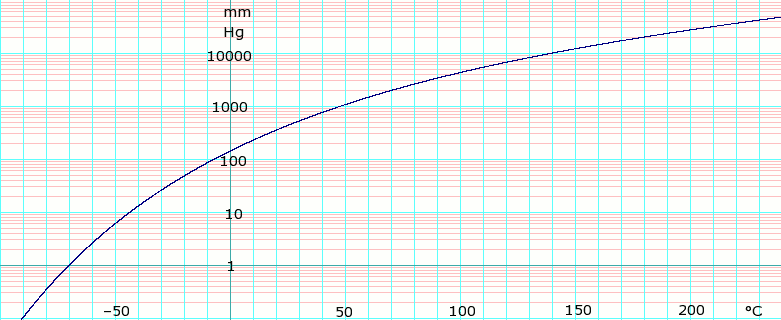
[[Image:LogMethylenechlorideVaporPressure.png|thumb|781px|left|'''log<sub>10</sub> of Dichloromethane vapor pressure.''' Uses formula: <math>\scriptstyle \log_e P_{mmHg} =</math><math>\scriptstyle \log_e (\frac {760} {101.325}) - 10.08632\log_e(T+273.15) - \frac {6030.610} {T+273.15} + 80.87786 + 9.812512 \times 10^{-6} (T+273.15)^2</math> obtained from CHERIC<ref name="cheric_p"/>]]{{Clear}} | |||
==Spectral data== | |||
{| border="1" cellspacing="0" cellpadding="3" style="margin: 0 0 0 0.5em; background: #FFFFFF; border-color: #C0C090;" | |||
! width="85%" align="center" cellspacing="3" style="font-size:95%; border: 1px solid #C0C090; background-color: #F8EABA; margin-bottom: 3px;" colspan="2" | [[UV/VIS spectroscopy|UV-Vis]] | |||
|- | |||
| Spectrum | |||
| ? | |||
|- | |||
| [[Lambda-max]] | |||
| ? [[Nanometre|nm]] | |||
|- | |||
| Log [[Extinction coefficient|Ε]] | |||
| ? | |||
|- | |||
! {{chembox header}} colspan="2" | [[Infrared|IR]] | |||
|- | |||
| Spectrum | |||
| [http://webbook.nist.gov/cgi/cbook.cgi?ID=C75092&Units=SI&Type=IR-SPEC&Index=2#IR-SPEC NIST] | |||
|- | |||
| Major absorption bands | |||
| ? cm<sup>−1</sup> | |||
|- | |||
! {{chembox header}} colspan="2" | [[NMR Spectroscopy|NMR]] | |||
|- | |||
| [[Proton NMR]] <!-- Link to image of spectrum --> | |||
| δ CDCl<sub>3</sub> 5.30 (s, 2H) | |||
|- | |||
| [[Carbon-13 NMR]] <!-- Link to image of spectrum --> | |||
| δ CDCl<sub>3</sub> 53.5 | |||
|- | |||
| Other NMR data <!-- Insert special data e.g. <sup>19</sup>F chem. shifts, omit if not used --> | |||
| ? | |||
|- | |||
! width="85%" align="center" cellspacing="3" style="font-size:95%; border: 1px solid #C0C090; background-color: #F8EABA; margin-bottom: 3px;" colspan="2" | [[Mass Spectrometry|MS]] | |||
|- | |||
| Masses of <br>main fragments | |||
| ? <!-- Give list of major fragments --> | |||
|- | |||
| {{chembox header}} colspan="2" | <small>Except where noted otherwise, data are given for<br>[[standard ambient temperature and pressure|standard ambient temperature and pressure (25°C, 101.3 kPa)]]<br/>[[wikipedia:Chemical infobox|Disclaimer and references]]</small> | |||
|} | |||
==Structure and properties data== | |||
{| border="1" cellspacing="0" cellpadding="3" style="margin: 0 0 0 0.5em; background: #FFFFFF; border-color: #C0C090;" | |||
! width="85%" align="center" cellspacing="3" style="font-size:95%; border: 1px solid #C0C090; background-color: #F8EABA; margin-bottom: 3px;" colspan="2" | Structure and properties | |||
|- | |||
| [[Index of refraction]] | |||
| 1.424 | |||
|- | |||
| [[Dielectric constant]] | |||
| 8.93 | |||
|- | |||
| [[Viscosity]] | |||
| 0.44 [[Poise|cP]] at 20 °C | |||
|- | |||
| width="85%" align="center" cellspacing="3" style="font-size:95%; border: 1px solid #C0C090; background-color: #F8EABA; margin-bottom: 3px;" colspan="2" | <small>Except where noted otherwise, data are given for<br>[[standard ambient temperature and pressure|standard ambient temperature and pressure (25°C, 101.3 kPa)]]<br/>[[wikipedia:Chemical infobox|Disclaimer and references]]</small> | |||
|} | |||
==References== | |||
{{reflist}} | |||
* [http://webbook.nist.gov/cgi/cbook.cgi?Name=dichloromethane&Units=SI NIST website] | |||
* G. W. C. Kaye and [[T. H. Laby]], [http://www.kayelaby.npl.co.uk/toc/ Tables of Physical & Chemical Constants at National Physical Laboratory] | |||
* [http://webbook.nist.gov/cgi/cbook.cgi?ID=C75092&Units=SI&Mask=3&Type=JANAFG&Table=on#JANAFG Heat capacity] | |||
Except where noted otherwise, data relate to [[standard ambient temperature and pressure]]. | |||
[[wikipedia:Chemical infobox|Disclaimer]] applies. | |||
{{DEFAULTSORT:Dichloromethane (Data Page)}} | |||
[[Category:Chemical data pages]] | |||
Revision as of 01:15, 18 January 2014
30 year-old Entertainer or Range Artist Wesley from Drumheller, really loves vehicle, property developers properties for sale in singapore singapore and horse racing. Finds inspiration by traveling to Works of Antoni Gaudí. Please find below supplementary chemical data about dichloromethane.
MSDS sheets
The handling of this chemical may incur notable safety precautions. It is highly recommend that you seek the Material Safety Datasheet (MSDS) for this chemical from a reliable source and follow its directions.
- SIRITemplate:Dead link
- Science Stuff
- Baker
- Oxford University Chemical Safety ArchiveTemplate:Dead link
Structure and properties
| Structure and properties | |
|---|---|
| Index of refraction,[1] nD | 1.4242 |
| Abbe number | ? |
| Dielectric constant,[2] εr | 9.08 ε0 at 20°C |
| Dipole moment,[3] | 1.14 D |
| Bond strength | ? |
| Bond length | ? |
| Bond angle | ? |
| Magnetic susceptibility | ? |
| Surface tension[4] | 26.52 dyn/cm at 20°C |
| Viscosity[5] | 0.449 mPa·s at 15°C 0.393 mPa·s at 30°C |
Thermodynamic properties
| Phase behavior | |
|---|---|
| Triple point | ? K (? °C), ? Pa |
| Critical point[6] | 510 K (237 °C), 6100 kPa |
| Std enthalpy change of fusion, ΔfusH |
+6.160 kJ/mol |
| Std entropy change of fusion, ΔfusS |
? J/(mol·K) |
| Std enthalpy change of vaporization, ΔvapH |
+28.6 kJ/mol |
| Std entropy change of vaporization, ΔvapS |
? J/(mol·K) |
| Solid properties | |
| Std enthalpy change of formation, ΔfH |
? kJ/mol |
| Standard molar entropy, S |
? J/(mol K) |
| Heat capacity, cp | ? J/(mol K) |
| Liquid properties | |
| Std enthalpy change of formation, ΔfH |
−124.3 kJ/mol |
| Standard molar entropy, S |
174.5 J/(mol K) |
| Heat capacity, cp | 102.3 J/(mol K) |
| Gas properties | |
| Std enthalpy change of formation, ΔfH |
−95.52 kJ/mol |
| Standard molar entropy, S |
270.28 J/(mol K) |
| Heat capacity, cp | ? J/(mol K) |
| van der Waals' constants[7] | a = 1244 L2 kPa/mol2 b = 0.08689 liter per mole |
Vapor pressure of liquid
| P in mm Hg | 1 | 10 | 40 | 100 | 400 | 760 |
| T in °C | –70.0 | –43.3 | –22.3 | –6.3 | 24.1 | 40.7 |
Table data obtained from CRC Handbook of Chemistry and Physics 47th ed.
50 year old Petroleum Engineer Kull from Dawson Creek, spends time with interests such as house brewing, property developers in singapore condo launch and camping. Discovers the beauty in planing a trip to places around the entire world, recently only coming back from .
Spectral data
| UV-Vis | |
|---|---|
| Spectrum | ? |
| Lambda-max | ? nm |
| Log Ε | ? |
| IR | |
| Spectrum | NIST |
| Major absorption bands | ? cm−1 |
| NMR | |
| Proton NMR | δ CDCl3 5.30 (s, 2H) |
| Carbon-13 NMR | δ CDCl3 53.5 |
| Other NMR data | ? |
| MS | |
| Masses of main fragments |
? |
| Except where noted otherwise, data are given for standard ambient temperature and pressure (25°C, 101.3 kPa) Disclaimer and references | |
Structure and properties data
| Structure and properties | |
|---|---|
| Index of refraction | 1.424 |
| Dielectric constant | 8.93 |
| Viscosity | 0.44 cP at 20 °C |
| Except where noted otherwise, data are given for standard ambient temperature and pressure (25°C, 101.3 kPa) Disclaimer and references | |
References
43 year old Petroleum Engineer Harry from Deep River, usually spends time with hobbies and interests like renting movies, property developers in singapore new condominium and vehicle racing. Constantly enjoys going to destinations like Camino Real de Tierra Adentro.
- NIST website
- G. W. C. Kaye and T. H. Laby, Tables of Physical & Chemical Constants at National Physical Laboratory
- Heat capacity
Except where noted otherwise, data relate to standard ambient temperature and pressure.
Disclaimer applies.
- ↑ Template:Cite web
- ↑ CRC Handbook of Chemistry and Physics, 47th ed. p E-54
- ↑ http://macro.lsu.edu/howto/solvents/Dipole%20Moment.htm
- ↑ CRC Handbook of Chemistry and Physics, 47th ed. pp F-28 - F-30
- ↑ CRC Handbook of Chemistry and Physics, 47th ed. pp F-33 - F-38
- ↑ 6.0 6.1 Template:Cite web
- ↑ Template:Cite web

